- Congreve, M., Langmead, C., Mason, J. and Marshall, F. (2011). Progress in Structure Based Drug Design for G Protein-Coupled Receptors. Journal of Medicinal Chemistry, 54(13), pp.4283-4311.
- de Opakua, A., Parag-Sharma, K., DiGiacomo, V., Merino, N., Leyme, A., Marivin, A., Villate, M., Nguyen, L., de la Cruz-Morcillo, M., Blanco-Canosa, J., Ramachandran, S., Baillie, G., Cerione, R., Blanco, F. and Garcia-Marcos, M. (2017). Molecular mechanism of Gαi activation by non-GPCR proteins with a Gα-Binding and Activating motif. Nature Communications, 8, p.15163.
- Du, C. and Xie, X. (2012). G protein-coupled receptors as therapeutic targets for multiple sclerosis. Cell Research, 22(7), pp.1108-1128.
- Dunworth, W. and Caron, K. (2009). G Protein-Coupled Receptors as Potential Drug Targets for Lymphangiogenesis and Lymphatic Vascular Diseases. Arteriosclerosis, Thrombosis, and Vascular Biology, 29(5), pp.650-656.
- Dunworth, W.P. and Caron, K.M., 2009. G protein–coupled receptors as potential drug targets for lymphangiogenesis and lymphatic vascular diseases. Arteriosclerosis, thrombosis, and vascular biology, 29(5), pp.650-656.
- Ghanemi, A. (2015). Targeting G protein coupled receptor-related pathways as emerging molecular therapies. Saudi Pharmaceutical Journal, 23(2), pp.115-129.
- Holliday, N., Watson, S. and Brown, A. (2012). Drug Discovery Opportunities and Challenges at G Protein Coupled Receptors for Long Chain Free Fatty Acids. Frontiers in Endocrinology, 2.
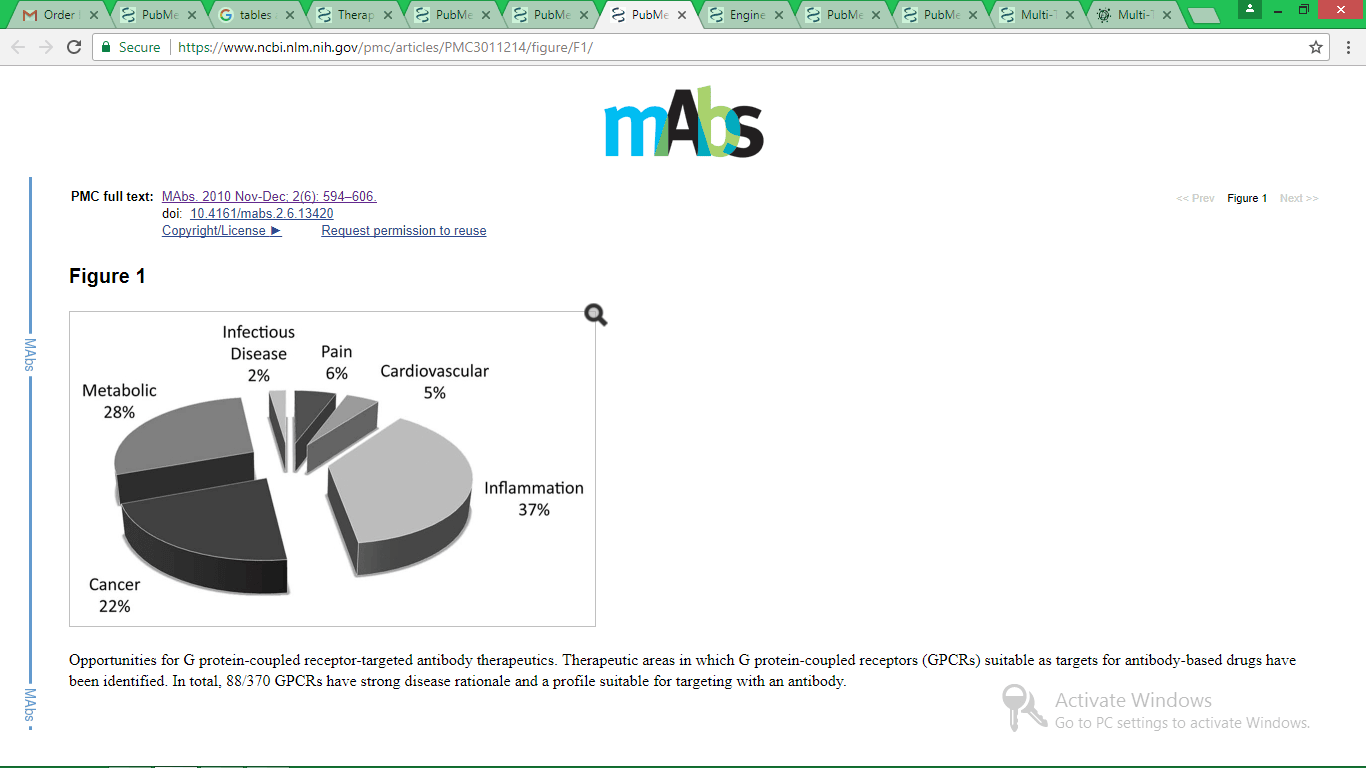
- Hutchings, C., Koglin, M., Olson, W. and Marshall, F. (2017). Opportunities for therapeutic antibodies directed at G-protein-coupled receptors. Nature Reviews Drug Discovery, 16(11), pp.787-810.
- Insel, P., Wilderman, A., Zambon, A., Snead, A., Murray, F., Aroonsakool, N., McDonald, D., Zhou, S., McCann, T., Zhang, L., Sriram, K., Chinn, A., Michkov, A., Lynch, R., Overland, A. and Corriden, R. (2015). G Protein-Coupled Receptor (GPCR) Expression in Native Cells: “Novel” endoGPCRs as Physiologic Regulators and Therapeutic Targets. Molecular Pharmacology, 88(1), pp.181-187.
- Jiang, M. and Bajpayee, N. (2009). Molecular Mechanisms of Go Signaling. Neurosignals, 17(1), pp.23-41.
- Kontoyianni, M. (2014). G Protein Coupled Receptors: Druggability and Structural Aspects. Journal of Autacoids, 05(01).
- Lappano, R. and Maggiolini, M. (2011). G protein-coupled receptors: novel targets for drug discovery in cancer. Nature Reviews Drug Discovery, 10(1), pp.47-60.
- Lundstrom, K. (2006). Latest development in drug discovery on G protein-coupled receptors. Curr Protein Pept. Sci., 7(5), pp. 465-70.
- Lundstrom, K. (2009). An Overview on GPCRs and Drug Discovery: Structure-Based Drug Design and Structural Biology on GPCRs. Methods in Molecular Biology, pp.51-66.
- Lundstrom, K. (2016). New winds in GPCR-based drug discovery. Future Medicinal Chemistry, 8(6), pp.605-608.
- McNeely, P., Naranjo, A. and Robinson, A. (2012). Structure-function studies with G protein-coupled receptors as a paradigm for improving drug discovery and development of therapeutics. Biotechnology Journal, 7(12), pp.1451-1461.
- McNeely, P.M., Naranjo, A.N. and Robinson, A.S., 2012. Structure‐function studies with G protein‐coupled receptors as a paradigm for improving drug discovery and development of therapeutics. Biotechnology journal, 7(12), pp.1451-1461.
- Miao, Y. and McCammon, J. (2017). Enhanced Simulations and Drug Discovery of a Muscarinic G-Protein-Coupled Receptor. Biophysical Journal, 112(3), pp.328a-329a.
- New, D. and Wong, Y. (2007). Molecular mechanisms mediating the G protein-coupled receptor regulation of cell cycle progression. Journal of Molecular Signaling, 2, p.2.
- Reimann, F. and Gribble, F. (2015). G protein-coupled receptors as new therapeutic targets for type 2 diabetes. Diabetologia, 59(2), pp.229-233.
- Reimann, F. and Gribble, F. (2015). G protein-coupled receptors as new therapeutic targets for type 2 diabetes. Diabetologia, 59(2), pp.229-233.
- Research Features. (2017). Challenges and solutions: G protein-coupled receptors as novel drug targets. Retrieved from: http://researchfeatures.com/2017/06/19/challenges-and-solutions-g-protein-coupled-receptors-as-novel-drug-targets/
- Salon, J., Lodowski, D. and Palczewski, K. (2011). The Significance of G Protein-Coupled Receptor Crystallography for Drug Discovery. Pharmacological Reviews, 63(4), pp.901-937.
- Salon, J.A., Lodowski, D.T. and Palczewski, K., 2011. The significance of G protein-coupled receptor crystallography for drug discovery. Pharmacological reviews, 63(4), pp.901-937.
- Schalop, L. and Allen, J. (2017). GPCRs, Desirable Therapeutic Targets in Oncology. Retrieved from: https://www.biosciencetechnology.com/article/2017/01/gpcrs-desirable-therapeutic-targets-oncology
- Schirone, L., Forte, M., Palmerio, S., Yee, D., Nocella, C., Angelini, F., Pagano, F., Schiavon, S., Bordin, A., Carrizzo, A., Vecchione, C., Valenti, V., Chimenti, I., De Falco, E., Sciarretta, S. and Frati, G. (2017). A Review of the Molecular Mechanisms Underlying the Development and Progression of Cardiac Remodeling. Oxidative Medicine and Cellular Longevity, 2017, pp.1-16.
- Seifert, R. (2005). G Protein-Coupled Receptors as Drug Targets: Analysis of Activation and Constitutive Activity.
- Smith, N. (2015). Drug Discovery Opportunities at the Endothelin B Receptor-Related Orphan G Protein-Coupled Receptors, GPR37 and GPR37L1. Frontiers in Pharmacology, 6.
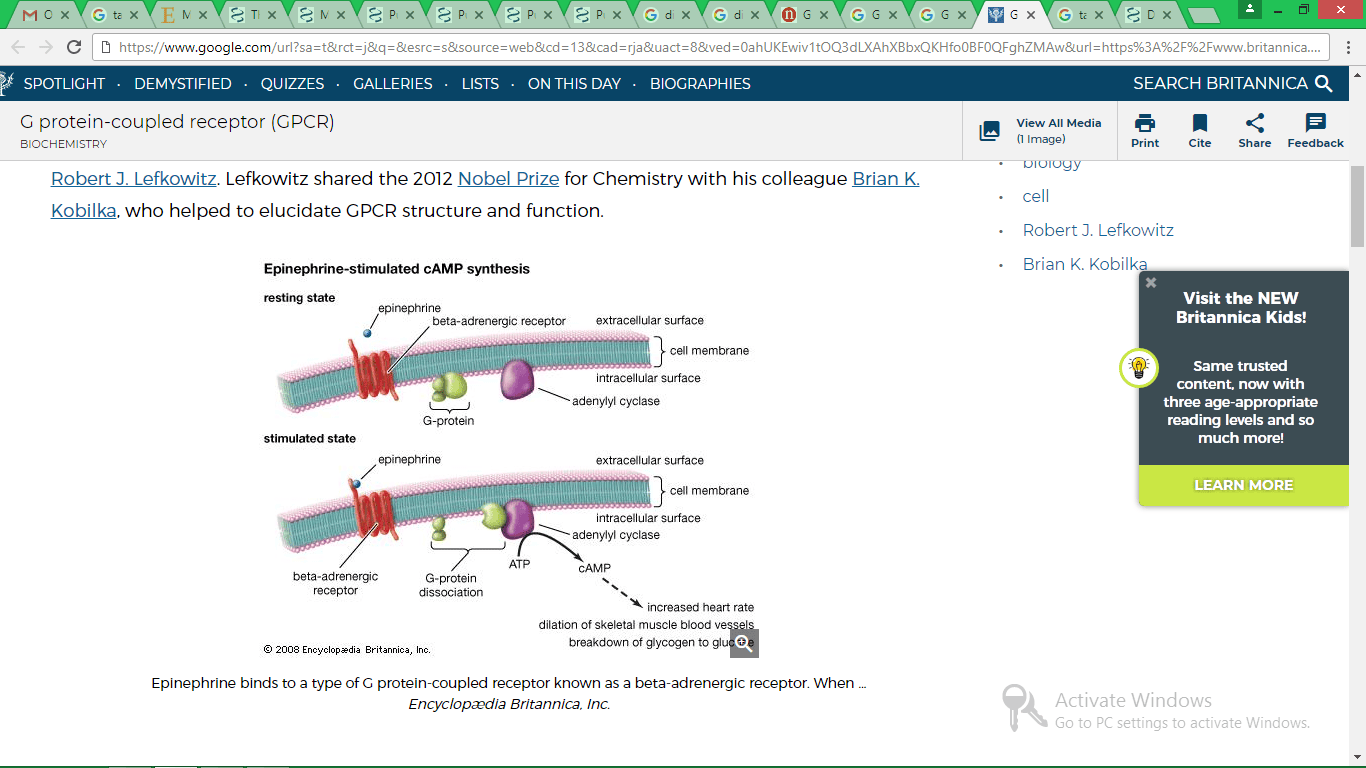
- Tuteja, N. (2009). Signaling through G protein coupled receptors. Plant Signaling & Behavior, 4(10), pp. 942–947
- Venkatakrishnan, A., Deupi, X., Lebon, G., Tate, C., Schertler, G. and Babu, M. (2013). Molecular signatures of G-protein-coupled receptors. Nature, 494(7436), pp.185-194.
- Zhang, Y., Scoumanne, A. and Chen, X. (2010). G Protein-Coupled Receptor 87: a Promising Opportunity for Cancer Drug Discovery. Mol Cell Pharmacol, 2(3): 111–116.
- Zhang, Y., Scoumanne, A. and Chen, X. (2010). G Protein-Coupled Receptor 87: a Promising Opportunity for Cancer Drug Discovery. Molecular and Cellular Pharmacology. 2(3), pp.111-116]
G-protein Coupled Receptors (GPCR) as Drug Targets
| Subject: | Science |
| Type: | Evaluation Essay |
| Pages: | 7 |
| Word count: | 1871 |
| Topics: | Biology, Cancer, Chemistry, Health, Innovation, Medicine |
Sources
More samples
Related Essays
Get a brand-new guide on essay writing
The download will start within seconds. Good luck!
Download Sample
This essay is publicly available.
Offered for reference purposes only.
Offered for reference purposes only.
By clicking Get this Sample you agree to the Terms And conditions and Privacy policy.
Thank you!
The download will start shortly.